Tackling Brain Disorders with Gene Therapy
A New Kind of Therapy
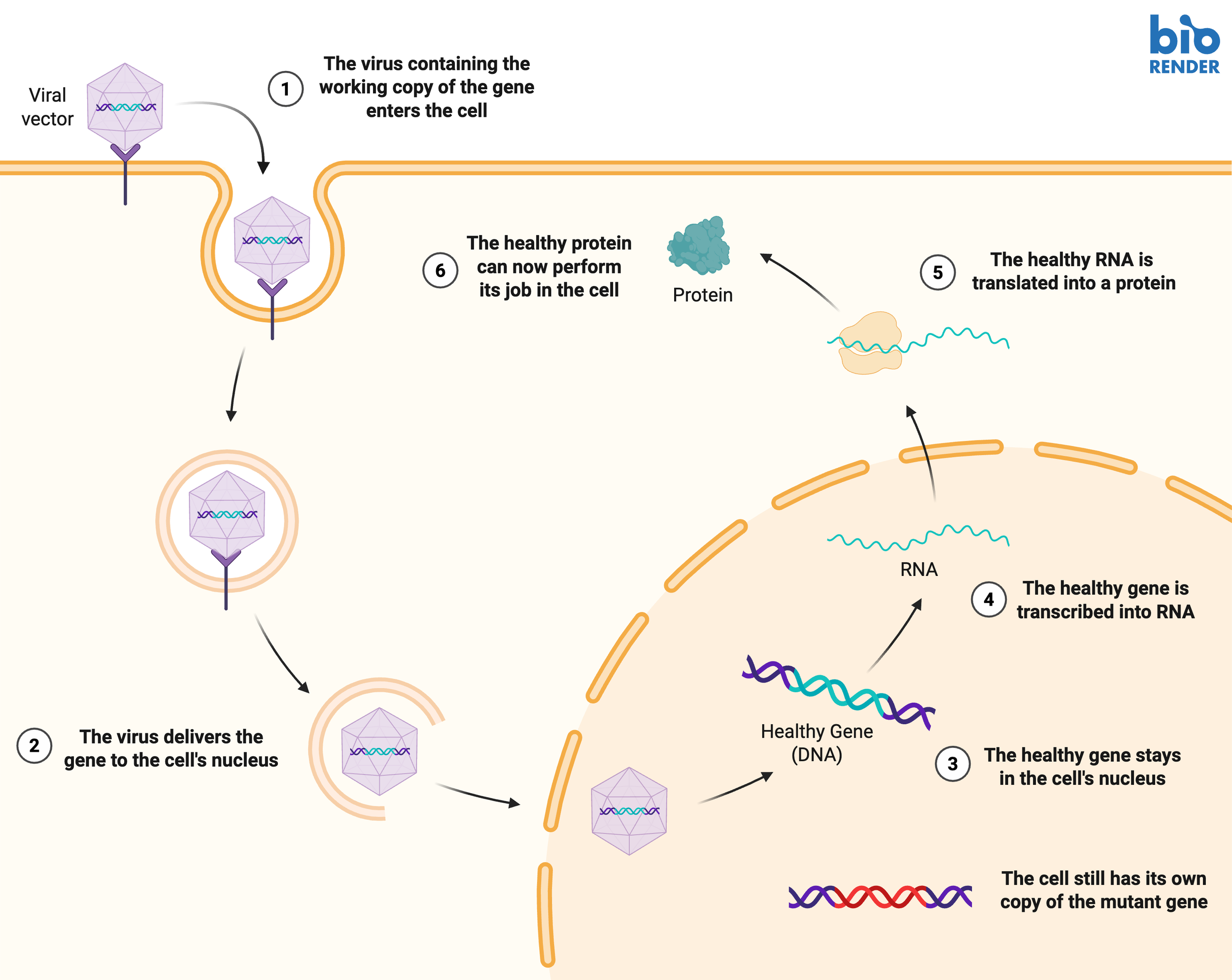
DNA serves as the instruction manual for our cells. It gets copied into RNA, which acts as a temporary “print out” of the instructions, in a process called transcription. Finally, proteins are made from RNA in a process called translation. Proteins act like mini machines that perform all the necessary tasks to keep cells healthy and functional. So what happens when the DNA instruction manual has a typo?
Genetic mutations are these typos, and they can lead to a range of disorders by creating faulty proteins or unstable RNA that can’t get translated into enough protein for normal functioning. Historically, many medications have targeted the end product – the proteins – to either modify the disease-associated protein or boost other parts of the pathway that the protein plays a role in. Gene therapy is a new way to treat genetic disorders by modifying or replacing the specific part of the DNA responsible – a mutant gene. Gene therapies hold great promise in curing otherwise devastating conditions, including those that affect the brain. While many therapies are being designed and tested, here are two examples which have already been approved by the FDA.
Replacing Faulty Genes
Spinal muscular atrophy, or SMA, is a rare genetic disorder that affects motor neurons [1]. People with SMA have mutations in the survival motor neuron 1 gene, called SMN1, which lead to too little SMN1 protein being produced. Motor neurons need this protein to function properly and to send signals to muscles to make the body move. Without enough SMN1, motor neurons die, and patients’ muscles become weakened. Depending on the severity of the mutation, SMA can lead to death even in infancy.
In 2019, the FDA approved Zolgensma, a one-time injection for patients under 2 years old [2,3]. This therapy introduces a new, working copy of the SMN1 gene to the patients’ motor neurons by packaging it into a virus delivery system. Rather than causing a disease, the virus has been modified to act like a delivery truck, able to enter cells to deliver their gene therapy package. The cell’s natural RNA-making machinery can read the new copy’s instructions, thus reinforcing the protein level of SMN1. This treatment has been highly effective in improving outcomes such as sitting independently and walking. Just last year, a similar drug Itvisma was approved for older patients with SMA4.
Editing Patients’ Own Cells
X-linked adrenoleukodystrophy (ALD) is caused by mutations in the ABCD1 gene [5]. ABCD1 codes for a protein that breaks down toxic buildup of certain fatty acids. Because ABCD1 is located on the X chromosome, mutations can affect males and females quite differently. Females have two copies of the X chromosome, so they can have a healthy backup copy of ABCD1. Males, however, only have one X and one Y chromosome. While most females with ABCD1 mutations only experience mild movement and bladder issues later in life, males may experience a much earlier decline in function in the brain and adrenal glands (organs that produce hormones) [6]. The most severe form is called cerebral ALD, where neurological symptoms progress to a point that patients die within a few years [5,6]. However, a gene therapy treatment is available, called Skysona [8]. It works by collecting patients’ own blood cells, which are given a working copy of ABCD1 using another viral delivery system. The healthy cells are then replaced back into the patient, where they divide and make more of the protein that breaks down the fatty acids. This helps to slow down the damage in the brain and increase chances of survival.
Gene Therapy Limitations
While great advancements have been made with gene therapy, there are still limitations to how effective or how available they are. Different kinds of mutations can exist in the same disease-related gene, so not every mutation can be helped by available gene therapies. Co-occurring conditions can make patients ineligible for some treatments. Long term side effects can pose great risks: for example, in blood cell transplant methods like Skysona, patients need to be carefully monitored for developing blood cancer for the rest of their life [7]. Gene therapies are often incredibly expensive: they can be millions of dollars per single treatment [8]. Lastly, though many disorders exist that could be cured with gene therapy, most drugs take years to be developed and to be deemed safe enough to be prescribed. Ultimately, gene therapies are just one of many tools for tackling complex genetic disorders.
References
Spinal muscular atrophy | National Institute of Neurological Disorders and stroke. (n.d.). https://www.ninds.nih.gov/health-information/disorders/spinal-muscular-atrophy
Mendell, J. R., Al-Zaidy, S., Shell, R., Arnold, W. D., Rodino-Klapac, L. R., Prior, T. W., Lowes, L., Alfano, L., Berry, K., Church, K., Kissel, J. T., Nagendran, S., L’Italien, J., Sproule, D. M., Wells, C., Cardenas, J. A., Heitzer, M. D., Kaspar, A., Corcoran, S., . . . Kaspar, B. K. (2017). Single-Dose Gene-Replacement therapy for spinal muscular atrophy. New England Journal of Medicine, 377(18), 1713–1722. https://doi.org/10.1056/nejmoa1706198
About ZOLGENSMA | ZOLGENSMA® (onasemnogene abeparvovec-xioi). (n.d.). https://www.zolgensma-hcp.com/about-zolgensma/about-zolgensma
Novartis. (n.d.). Home | ITVISMA®. https://www.itvisma.com/
Scott, A. I., Mallhi, K. K., Ganesh, J., Chen, W. L., & ACMG Therapeutics Committee8∗documents@acmg.net (2023). Elivaldogene autotemcel approved for treatment of cerebral adrenoleukodystrophy (CALD) in males: A therapeutics bulletin of the American College of Medical Genetics and Genomics (ACMG). Genetics in medicine open, 1(1), 100835. https://doi.org/10.1016/j.gimo.2023.100835
Adrenoleukodystrophy (ALD) | New York State Department of Health, Wadsworth Center. (n.d.). https://www.wadsworth.org/public-health-programs/newborn-screening/newborn-screening-program/adrenoleukodystrophy-ald
Musolino, P. L., Gong, Y., Snyder, J. M., Jimenez, S., Lok, J., Lo, E. H., Moser, A. B., Grabowski, E. F., Frosch, M. P., & Eichler, F. S. (2015). Brain endothelial dysfunction in cerebral adrenoleukodystrophy. Brain : a journal of neurology, 138(Pt 11), 3206–3220. https://doi.org/10.1093/brain/awv250
SKYSONA™ (elivaldogene autotemcel) | An FDA Approved Gene Therapy. (n.d.). https://www.skysona.com/
Wong, C. H., Li, D., Wang, N., Gruber, J., Conti. R. M., and Lo, A. W. (2021) Estimating the Financial Impact of Gene Therapy in the U.S. NBER Working Paper 28628. https://doi.org/10.3386/w28628.
Gene therapy illustration created by Aleta Murphy in BioRender
Thumbnail by Anirudh on Unsplash
Edited by Emma Hays